Highly stable under hydrogen sulfide exposure environment without sulfation or discoloration, with stable battery performance and durability during charging and discharging.

Developed materials (electrolytic iron foil/Fe-Ni alloy foil)


Tensile strength (copper foil vs. ferrous metal foil)
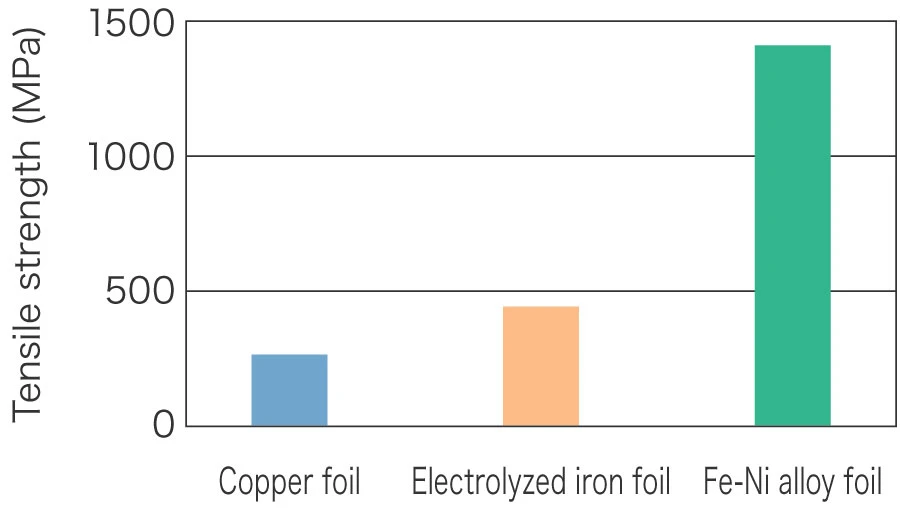
Characteristics
Gas corrosion test (hydrogen sulfide)

Contact resistance ratio: 3.2

Contact resistance ratio: 1.0

Contact resistance ratio: 1.0
This is a before/after test comparison.
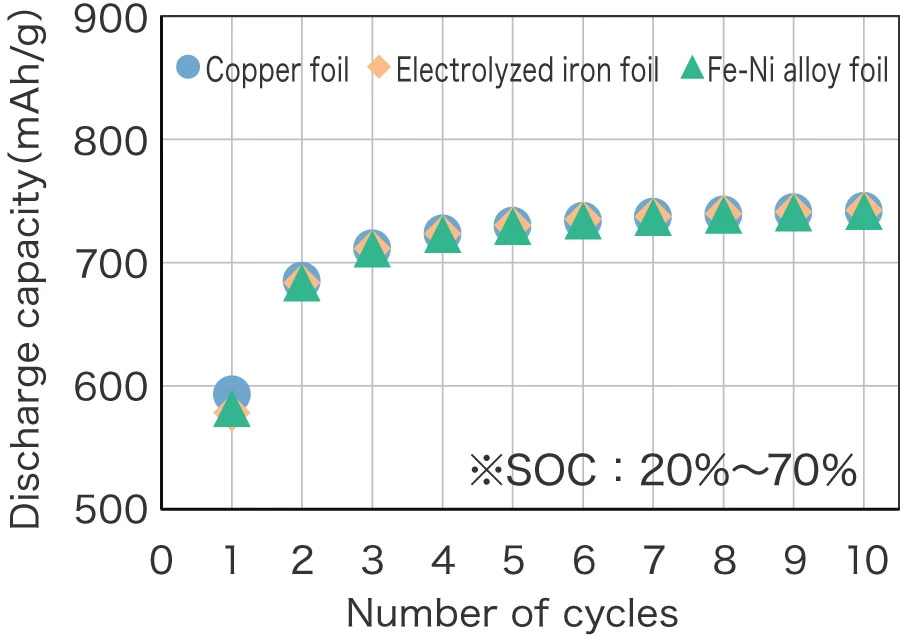
Cycle characteristics of SiO anode using various current collectors
External image of test battery after dismantling

(10µm)

(10µm)

(10µm)
Joint research organization: AIST Battery System

